Data – What we need to do the analysis? But how much we are sure that the data we have collected for analysis is reliable enough! The main agenda of this post is to discuss data reliability, it’s importance and how to achieve it.
Let’s start with a brief example, suppose during a Clinical Trial “If the data are not taken properly?” And proceed further for analysis. Then “What will be the consequences of it?” Clearly, it may fail later and results in loss of lives, resources and time. Thus, the device measuring the host condition should produce reliable results.
Now, I would like to introduce the term called “Measurement System Analysis (MSA)” which helps to determine the reliability of a data for analysis. You must be curious to know about it, right!
Measurement system in Healthcare
Generally, MSA is a method to determine the measurement system in a process. The measurement system is a collection of measurement devices, measurement procedures and operators that are used to obtain a measurement. MSA helps us to detect the variation (i.e difference or disparity) exists in a measurement system. So we should evaluate our measurement system before conducting statistical process control, design of experiment, Predictive Analytics, or other statistical analysis. By doing so, we can be sure that our data are reliable for analysis.
We can apply MSA for two types of data namely attribute and continuous data.
- Attribute data - When data collected are in the form of count or categorized in the form of group. For e.g. accept or reject, good or bad, and so on. In such scenario, we use Attribute agreement analysis.
- Continuous data - When data collected are in the form of fractional or decimal value. For e.g. weight, length, time, temperature, etc. In such scenario, we use a Gage R & R analysis.
MSA is classified into Attribute agreement analysis and Gage R&R analysis based on data types used.
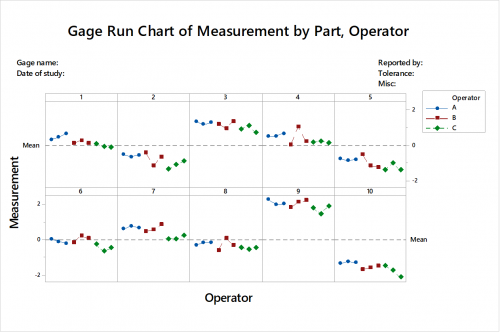
Gage Run Chart
Example
Suppose in a Wheel Manufacturing Company, operators use gage to measure the diameter of the wheel. Are we 100% sure that the collected data is measured correctly or not?
With reference from the Gage Run Chart – Three operators measure 10 wheels, three times per wheel randomly. Here, we can examine the differences in measurements between operators. We can see that Operator B does not measure consistently, and Operator C usually measures lower than the other operators.
Let’s discuss a few terminologies used in measurement system analysis.
- Accuracy – It is the difference between the measurement of a part and its actual value.
- Bias – It is the measurement difference between the observed value and true value.
- Linearity – It is the difference or a change in bias value w.r.t its size.
- Stability – It is a measurement of how well the system performs over time or we can say, it is a change in bias value over time.
- Precision – It is the closeness between the measured value and the true value.
- Repeatability – It is a variation which occurs when the same operator measures the same part with the same device.
- Reproducibility – It is a variation which occurs when the different operators measure the same part with the same device.
Why MSA is important?
It is a prerequisite for successfully implementing Lean Manufacturing, Six Sigma and Lean Six Sigma projects. It is also useful in the implementation of SPC, DOE, Enterprise Prediction Management, Enterprise Risk Management, Enterprise Risk Management, and Enterprise Quality Management processes. It gives the better reliability of the measurement system in a statistical way.
NB – Want to learn, How to run Measurement System Analysis in Minitab Software? Attend our Minitab Certified Training Program, starting from basic to advanced level. Some of the Minitab software training certified courses are Minitab Essentials, Statistical Tools for Pharmaceuticals, Statistical Quality Analysis & Factorial Designs, etc. Apart from Minitab training, we also conduct basic and advanced Statistical training. Some of the Statistical training certified courses are Predictive Analytics Masterclass, Essential Statistics For Business Analytics, SPC Masterclass, DOE Masterclass, etc.
We provide a wide range of Analytics & AI Solutions like Business Analytics, Digital Process Automation, Enterprise Information Management, Enterprise Decisions Management and Business Consulting Services for Organisations to enhance their decision support systems.
Related Posts

What role do t-tests play in Pharmaceutical Processes? When can we apply it?
First of all, I would like to set the concept of hypothesis testing then we will move step by step to the agenda….
- Oct 05

How Data Science can help to Improve the present Healthcare Scenarios?
According to the National Health profile 2018 released by Central Bureau of Health Intelligence it was reported..
- Oct 05
Categories
Recent Posts
- What role do t-tests play in Pharmaceutical Processes? When can we apply it?
- How Data Science can help to Improve the present Healthcare Scenarios?
- What can we Discover from the Process Data by Creating a Simple Histogram?
- What are the Quality Tools Available in Minitab?
- Why Choose Minitab as your Statistical Data Analytics Software?
Recent Comments